Maneja del Moko en banano
Manejo del Moko en banano con inductores de resistencia y dióxido de cloro
Joaquín Guillermo Ramírez G., Melissa Muñoz A., and Juan Gonzalo Morales O. (Departamento de Ciencias Agronómicas, Facultad de Ciencias Agrarias, Universidad Nacional de Colombia. Medellin. jgmoraleso@unal.edu.co)
Luis Fernando Patiño H. (Facultad de Ciencias Agrarias, Politécnico Colombiano Jaime Isaza Cadavid. Medellin.)
ABSTRACT
The plant disease Moko, caused by Ralstonia solanacearum, is the most important bacterial disease in banana and plantain crops worldwide. In the present study, chlorine dioxide and seven resistance inducers in banana plants (Musa sp.) infected with this bacterium were evaluated under greenhouse condi- tions. For the evaluation of chlorine dioxide, three doses were used (10, 30 and 50 mg L-1). The evaluation of the resistance inducers included the following: sodium salicylate 0.4 g L-1; hydrogen peroxide 1 mM; potassium phosphite 1.5 mL L-1; 3-aminobutanoic acid 1.0 g L-1; methyl jasmonate 0.2 g L-1; acibenzolar-s-methyl 0.3 mL L-1 and chitosan 3.0 mg mL-1. The results showed a significant reduction of 74% in the area under the disease progress curve (AUDPC) value, which was calcu- lated for the disease development when the injected chlorine dioxide dose was 50 mg L-1. The AUDPC value for the resistance inducers was reduced by 45.4% for chitosan, 75.5% for methyl jasmonate and 65.5% for 3-aminobutanoic acid. Therefore, the results indicated that these molecules have the potential to be used for control of the Moko disease.
RESUMEN
La enfermedad denominada Moko, ocasionada por Ralstonia solanacearum, es la principal enfermedad de origen bacteriano en los cultivos de banano y plátano en todo el mundo. Este trabajo tuvo como objetivo la evaluación del dióxido de cloro y siete inductores de resistencia en plantas de banano (Musa sp.) infectadas con la bacteria bajo condiciones de invernadero. Para la evaluación del dióxido de cloro se utilizaron tres dosis (10, 30, 50 mg L-1). Los inductores de resistencia utilizados y sus dosis fueron: salicilato de sodio: 0,4 g L-1, peróxido de hidrógeno: 1 mM, fosfito de K: 1,5 g L-1, ácido 3-aminobutanoico: 1,0 g L-1, metil jasmonato: 0,2 g L-1, acibenzolar-s-metil: 0,3 g L-1 y quitosano: 3,0 mg mL-1. Los resultados muestran una reducción significativa del 74% en el valor del área bajo la curva del progreso de la enfermedad (ABCPE), cuando se utiliza dióxido de cloro en dosis de 50 mg L-1 aplicado por inyección. Para los inductores de resistencia se observa una reducción de 45,4% para quitosano, 75,5% para metil jasmonato y de 65,5% para el ácido 3-aminobutanoico en el ABCPE. Por lo tanto, los resultados indican que estas moléculas tienen el potencial para ser usadas en el control del Moko.
INTRODUCTION
Banana (Musa sp.) is an important economic, social and nutritional crop in the countries where it is produced or imported. This fruit is grown in countries with tropical climates and it has a wide variety of consumption possibilities that include fresh or processed products available for national consumption or export (Roldán et al., 2002; Sena, 2004). Colombia exports bananas more than any other fruit; therefore, bananas represent an important asset to the Colombian economy, not only for its contribution to the gross domestic product but also because it generates direct and indirect jobs (Augura, 2012).
The production of bananas, globally and in Colombia, faces several limitations, and one such limitation is disease, including “Moko disease or bacterial wilt,” which is caused by Ralstonia solanacearum (Race 2) (Smith, 1993). In Colombia, this disease is important because of the ease of its dissemination, high aggressiveness, wide range of hosts, lack of effective management practices, high economic losses and eradication that requires long periods without cultivation or production. Additionally, misdiagnosis of the disease may lead to incorrect management (Buitrago, 2001; Arenas, 2004; Castañeda and Espinosa, 2005; Augura, 2012; Prieto et al., 2012).
New alternatives for the management of disease are constantly being researched, in particular for diseases of bacterial origin that exhibit difficult management (Agrios, 2005). Chlorine dioxide may be a novel option and it has already been used in the field; however, systematic stud- ies have not been performed that would provide scientific evidence of its benefits despite existing studies that have reported a reduction of the bacterial load in fruits and vegetables in the postharvest stage through the use of chlorine dioxide (Rodgers et al., 2004). On the other hand, induced resistance can be used against a wide spectrum of diseases through the use of a plant’s own resistance mechanisms and it has been widely studied (Herman et al., 2007; Walters, 2009). This induced resistance is considered to be variable, with levels of disease control ranging from 4% to more than 90% (Vallad and Goodman, 2004; Walters et al., 2005; Walters, 2009).
Because of the importance of the Moko disease (bacterial wilt) in the Musaceae family and the limited number of effective control strategies that currently exist, this study aimed to identify alternative strategies for the establishment of an integral disease management plan by evaluating several doses of chlorine dioxide and seven resistance inducers in the banana plant (Musa sp.) cultivar Williams infected with the bacterium R. solanacearum (Race 2) under greenhouse conditions.
Materials and methods
Location
The present study was performed in the greenhouse and Molecular Studies Laboratory of the Faculty of Agricultural Sciences in the Universidad Nacional de Colombia, Medellin (6o15’N, 75o35’W, 1,495 m a.s.l.).
Plant material
In-vitro banana seedlings cv. Williams (AAA) were obtained from the laboratory of Universidad Católica de Oriente, (Rionegro, Colombia). The seedlings were transferred to pots containing 3.5 kg of substrate (in a 3:1:1 ratio of soil, rice husk and quartz) that was previously autoclaved (103.42 kPa and 121oC for two cycles of 1 h each). The seedlings were kept under greenhouse conditions (mean temperature 22oC, relative mean humidity 85%, soil humidity 50% of the maximum humidity retention capacity and fertilization with Hoagland nutritive solution). The experiments started when the seedlings had 3 months of age.
Microorganisms
A strain of R. solanacearum (race 2) was provided by Centro de Investigaciones del Banano (CENIBANANO, Uraba- Colombia). The strain was replicated in the laboratory using a semi-selective culture medium, Kelman-TZC-Agar (K-TZC-A) (1 g 2,3,5-triphenyl-2H-tetrazolium chloride, 10 g dextrose, 10 g peptone, 1 g casamino acids and 18 g agar dissolved in 1 L H2O) (Kelman, 1954).
Pathogen inoculation
The inoculation process was conducted following the method of Obregon et al. (2008), in which pure colonies grown in an K-TZC-A medium (Kelman, 1954) were regrown in this same liquid medium (K-TZC) under cons- tant shaking (150 rpm) for 24 h at 25oC. The inoculation was performed using a 5 mL hypodermic syringe (Nipro, Osaka Japan) filled with a 2.5 mL solution containing an approximate concentration of 1·108 colony forming units (cfu) per mL, measured by spectrophotometry at 640 nm (NanoDrop 2000, spectrophotometer Thermo Scientific®, Wilmington, DE) (Smith, 1993; French et al., 1995).
Evaluated treatments
Chlorine dioxide
Phytotoxicity test on the banana seedlings. To analyze the phytotoxicity, the pseudostem of the seedlings at 12 weeks of age was injected at the lower end with 50, 30 and 10 mg L-1 chlorine dioxide using a hypodermic syringe. Each treatment was performed on five banana seedlings under greenhouse conditions and a control without treatment was included. Possible toxicity symptoms such as discoloration, sagging, abnormal growth, leaf burn and wilt and the height and diameter at the base of the stem were recorded weekly. Each treatment was applied weekly on the same plants for 3 weeks (days 0, 7 and 14).
In vitro inhibition test of chlorine dioxide on R. sola- nacearum. The test in a liquid medium was performed in 50 mL Falcon tubes containing 15 mL of K-TZC medium (Kelman, 1954) under shaking at 150 rpm and at 25-30oC. For the growth test in a solid media, five petri dishes for each chlorine dioxide concentration tested that contained 15 mL of K-TZC-A medium (Kelman, 1954) were placed under conditions of 12 h light and 12 h darkness at 25-30oC. In each medium type, five chlorine dioxide doses (0, 10, 30, 50 and 70 mg L-1) were evaluated for R. solanacearum. To analyze the effect of the different doses in the liquid media, the bacterial concentration was measured by spectropho- tometry (640 nm) at 3, 6, 9, 12, 18, 24, 30 and 36 h after inoculation. For the solid media, the effect of the different doses was evaluated using cfu counts over the 6 d after the start of the experiment.
Chlorine dioxide tests on the banana seedlings infected with R. solanacearum. Based on the in vitro evaluation and phytotoxicity tests, three chlorine dioxide doses (10, 30 and 50 mg L-1) were injected into the stem (I) and drenched (D) in the soil as previously described. The treatment was applied when the first external symptoms were visualized corresponding to the necrosis of the candle leaf, according to the disease rating scale reported by Silva et al. (2000).
Resistance inducers
Phytotoxicity test on the banana seedlings. To evaluate possible problems associated with high doses of inducers, phytotoxicity tests were performed for the banana seedling ‘Williams’. Symptoms such as discoloration, sagging, abnormal growth, leaf burn and wilt were evaluated and the height and diameter at the base of the stem were measured. The tests included two or three doses of each inducer applied to the leaves as follows: sodium salicylate (SaNa) (0.4, 0.8 and 1.2 g L-1), hydrogen peroxide (H2O2) (1.2 and 3.0 mM), potassium phosphite (FosK) (1.5, 2.0 and 2.5 mL L-1), 3-aminobutanoic acid (BABA) (1.0, 1.5 and 2.0 g L-1), methyl jasmonate (MeJa) (0.2, 0.4 and 0.6 g L-1), acibenzolar-s-methyl (ASM) (0.3, 0.6 and 0.9 mL L-1) and chitosan (Qt) (3.6 and 9.0 mg mL-1).
Effect of different resistance inducers on the banana seedlings infected with R. solanacearum. Based on the phytotoxicity tests, the following doses of resistance induc- ers were applied: SaNa 0.4 g L-1; H2O2 1.0 mM; FosK 1.5 mL L-1; BABA 1.0 g L-1; MeJa 0.2 g L-1; ASM 0.3 mL L-1; and Qt 3.0 mg mL-1. The products were applied on the leaves using a manual pressure sprayer (10.34 kPa) until run-off, 24 h prior to the inoculation and on the 10th d after the start of treatment.
Variables evaluated for the chlorine dioxide and resis- tance inducer experiments. For both experiments, the development of the disease was evaluated using the exter- nal symptoms rating scale reported by Silva et al. (2000), which establishes five stages: 1) no symptoms, 2) necrosis of the candle leaf, 3) yellowing of 2-3 leaves, 4) buckle of the petiole, 5) death of the plant. Based on the data obtained from the evaluations, the area under the disease progress curve (AUDPC) was calculated (Madden et al., 2007) and its dynamics were recorded over time. At the end of the experiment, the dry biomass was measured (dried in an oven at 60oC for 72 h). The data were recorded every 3 d until the inoculated controls reached stage 5 of the disease corresponding to death of the plants. At the end of the experiments, the presence of the pathogen was verified by dissecting the tissue and re-isolating it on K-TZC-A (Kelman, 1954).
Statistical analysis
A completely randomized experimental design was em- ployed, with one plant as the experimental unit and five replicates used for each treatment. Two replicates in time were performed for each experiment. The homoscedasticity and normality of the data were analyzed (P≤0.01) using the Levene and Kolmogorov-Smirnov tests, respectively. Subsequently, an analysis of variance (ANOVA) and mean separation test were performed using Tukey’s test with a significance level of 99%. The data were analyzed using the statistical software SAS® v.9.1.
Chlorine dioxide
Chlorine dioxide phytotoxicity test on the banana seedlings. No phytotoxicity symptoms were found when the seedlings were treated with all three chlorine dioxide doses (10, 30 and 50 mg L-1). In fact, the seedlings treated with chlorine dioxide had improved growth as compared to the control (P≤0.01), showing the higher values at 30 mg L-1 (Fig. 1A). Although Cl- is not essential to plants and is not required in high concentrations, it is considered beneficial because it may help with photosynthesis and water regulation in the plant (Rodgers et al., 2004). However, the dynamics of Cl- within the plant and dose applications in the field must be assessed so that chlorine dioxide may be correctly used with no adverse effects.
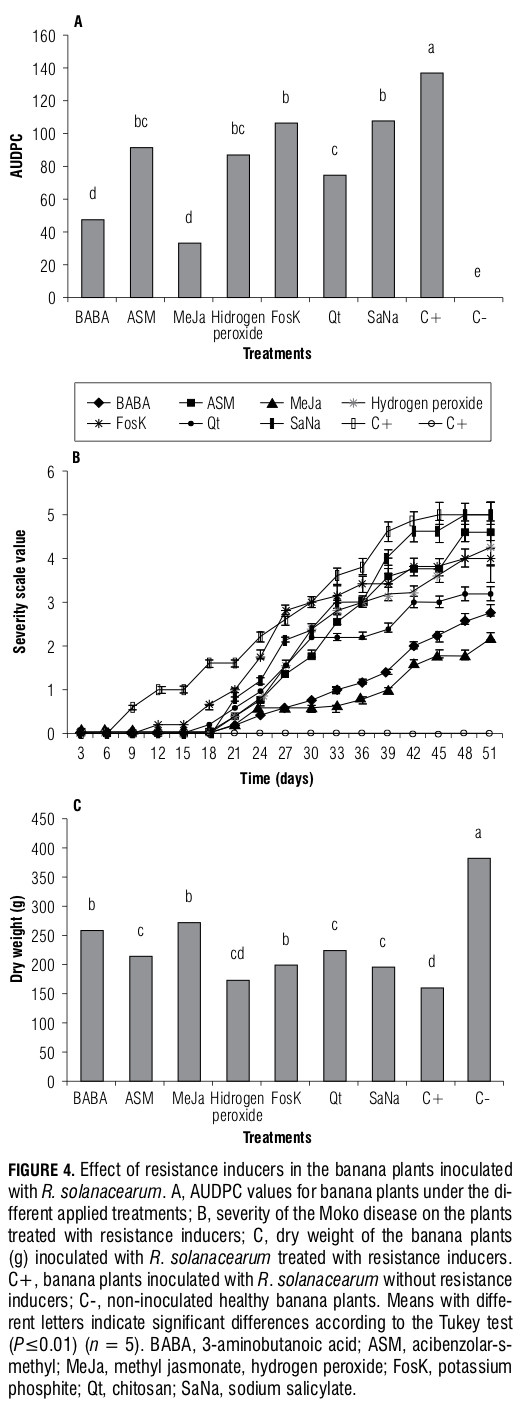
In vitro inhibition test of chlorine dioxide on R. sola- nacearum. The results obtained in the in vitro inhibition test in the liquid media showed that the treatments using 50 and 70 mg L-1 chlorine dioxide had an immediate bactericidal effect, because an increase in the optical density was not observed for these doses, as compared to the non-treated control, which may indicate that no bacterial growth occurred (Fig. 1B). However, the 10 and 30 mg L-1 doses had an inhibitory effect that was only limited to the 1st h of treatment. At 9 h after the start of the treatment, an increase in bacterial concentration was observed and these results were confirmed in the solid media test (Tab. 1); the 50 and 70 mg L-1 dose treatments showed no bacterial growth, which was indicated by the absence of cfus, whereas microbial growth was evident in the 30, 10 mg L-1 and control treatments (Fig. 1C).
The results suggested that chlorine dioxide is a highly reactive and efficient product in the initial stages of the plant-pathogen interaction, but if the correct dose is not delivered, the product cannot eliminate subsequent growth of the bacterial population. The action of chlorine dioxide on bacteria is considered to be direct and its chemical form allows it to penetrate the bacterial membrane and kill the organism (Junli et al., 1997).
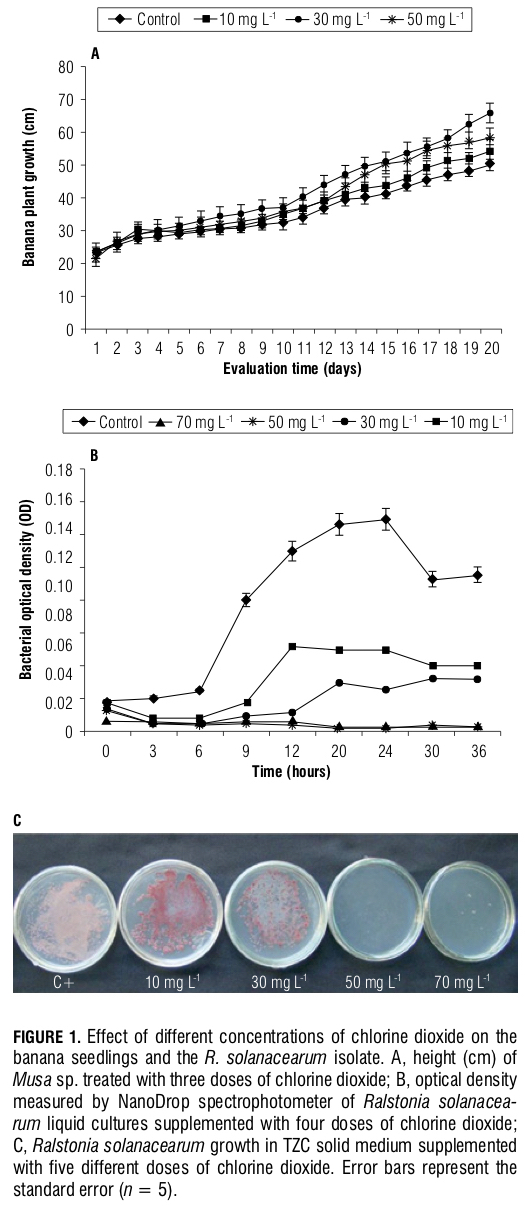
Chlorine dioxide tests on the banana seedlings infected with R. solanacearum. Significant differences (P≤0.01) were found between the treatments with respect to the AUDPC (Fig. 2A), with which Tukey’s test clustered the treatments into four different groups. The first group corresponded to the inoculated control (C+) that had the highest disease development. The second group corresponded to the 10 and 30 mg L-1 injections of chlorine dioxide and the 10, 30 and 50 mg L-1 chlorine dioxide drenched soil that had a medium-level AUDPC value. The third group corresponded to the 50 mg L-1 injections of chlorine dioxide and it had the lowest disease development. Finally, the last group was formed by the non-inoculated control, where no disease was found.
The inoculated control exhibited a significantly (P≤0.01) higher disease severity at the second week of evaluation than the other treatments (Fig. 2B). This same tendency occurred up to week five, where no statistically significant differences were found (P≤0.01) between the inoculated controls and plants subjected to the 10 and 30 mg L-1 chlorine dioxide drenched soil treatments. At week seven, the plants treated with chlorine dioxide at 10, 30 and 50 mg L-1 in drenched soil, 10 mg L-1 injections and inoculated controls showed no statistically significant differences (P≤0.01) in the dynamics of the disease severity, which was evaluated up to plant death. However, the 30 and 50 mg L-1 injections at the pseudostem showed that the speed of disease growth was significantly slower (P≤0.01), as compared with the other treatments. Here, the 50 mg L-1 chlorine dioxide injection in the stem showed the lowest response, confirming the data found for the AUDPC variable (Fig. 2A).
Regarding the total biomass variable (Fig. 2C), the inocu- lated control plants, plants treated with chlorine dioxide at 10 and 30 mg L-1 (both injected and in drenched soil) and plants treated with 50 mg L-1 drenched soil showed a significantly lower total biomass (P≤0.01) compared to the non-inoculated controls and 50 mg L-1 injection treatment. The 50 mg L-1 injection treatment achieved an increase in biomass despite the bacterial infection. The results obtained for the AUDPC, disease dynamics and biomass are visually confirmed in Fig. 3 A, B and C, where the best health stage can be observed in the plants injected with 50 mg L-1 chlorine dioxide at the pseudostem.
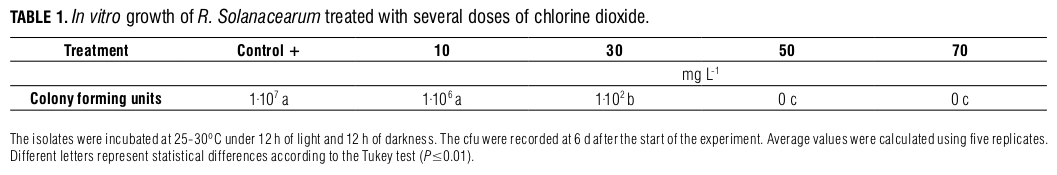

The results found in this study suggest a potential use for chlorine dioxide as a bactericidal agent when 50 mg L-1 is injected in the pseudostem of banana plants infected with R. solanacearum. This compound achieves a reduction in the disease development and improves a plant’s growth by increasing its biomass. Therefore, this product can be considered an alternative for the integral management of this crop; however, these results must be verified under field conditions to establish the efficacy of the treatment. In
addition, experiments with repeated doses over time must be performed to determine if the total eradication of the bacteria can be achieved in sick plants. Also, it is important to evaluate the treatment with drenched soil because, as this study demonstrated, this chlorine dioxide treatment was not particularly effective and its broad spectrum action can affect native bacterial populations in the soil, which might lead to an imbalance with negative consequences for the agroecosystem.
Studies reporting the direct action of chlorine dioxide in the management of plant diseases are scarce because, in most situations, this product has been evaluated in the post-harvest stage as a disinfectant, where it produces excellent results, such as those reported by Singh et al. (2002), who managed to reduce the bacterial load of Escherichia coli 0157:H7 in lettuce leaves using this compound. These results are consistent with the results reported by Zhang and Faber (1996), who demonstrated that 5 mg L-1 chlorine dioxide can eliminate a higher load of Listeria monocytogenes in lettuce leaves, as compared to water washing. However, Rodgers et al. (2004) reported that chlorine dioxide at 3 and 5 mg L-1 had a direct effect on Escherichia coli O157:H7 and Listeria monocytogenes in apple, lettuce and strawberry. In banana Ramirez et al. (2014) reported the use of Chlorine dioxide in doses of 10, 20 and 30 mg L-1 to reduce the incidence and severity of the soft rot of the pseudo-stem cause by Dickeya sp.
Evaluation of resistance inducers in plants
inoculated with R. solanacearum
Phytotoxicity test on the banana seedlings. The two higher doses of all of the resistance inducers used caused a degree of phytotoxicity that presented burned, curling and spotted leaves and delayed growth; therefore, the lower doses were used.
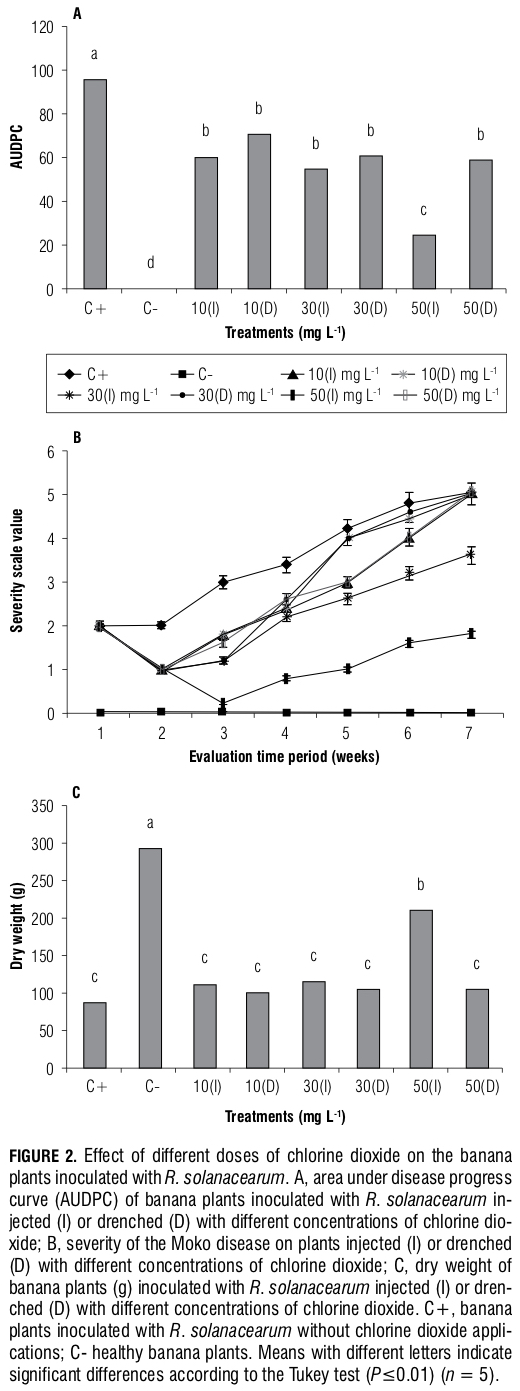
Effect of resistance inducers in the banana plants in- oculated with R. solanacearum. Based on the Tukey test, no statistically significant differences (P≤0.01) were found for the AUDPC of the plants that were treated with FosK, Sana, H2O2 and ASM; these inducers only managed to reduce the expression of the pathogen compared with that of the inoculated control (Fig. 4A). However, the Qt treatment showed a significant reduction (P≤0.01) in the disease compared with the inoculated control and the in- ducers SaNa and FosK. However, this reduction was lower than that of the MeJa and BABA inducers, which produced the highest decrease in the AUDPC (Fig. 4A). In the non- inoculated control plants, a pathogen development was not observed. The results obtained for the AUDPC variable

et al., 2004; Ozeretskovskaya et al., 2009; González et al., 2009; Falcón-Rodríguez et al., 2011). Regarding the Baba inducer and its effect on the reduction of R. solanacearum in banana, its action may be caused by its association with the accumulation of Pathogenesis-Related proteins in tissues treated before contact with the pathogen (Jakab et al., 2001; Siegrist et al., 2000). In practical studies, the BABA inducer has presented an effect on the control of Fusarium solani in potato (Olivieri et al., 2009; Yin et al., 2010). Also, the MeJa inducer has been the subject of fewer stud- ies and its effect is considered to be the result of the rapid induction of hydrogen peroxide (H2O2) during the early stages of the infection process (Zhu and Tian, 2012), which would explain its ability to decrease the pathogenic effect of R. solanacearum in inoculated banana plants.
It is important to understand that the action of resistance inducers is highly variable and depends on multiple factors (Walters, 2009). Therefore, the products that did not work under the conditions of this study cannot be discarded because, under different conditions, their effect may improve. Based on the results of this study, to define and promote the use of Qt, Meja and Baba in the control of R. solanacearum, the mechanisms by which these products achieve the observed effects must be identified. In addition, it is necessary to identify the basic aspects for their application in the field, including the correct dose and application frequency and their effects on growth and development, because these products require additional energy expenditures by the plant through the activation of several metabolic pathways (Walters, 2009). However, the effect of these products on sick plants could be analyzed without such energy expenditures because evaluations of the inducers in this study were performed 24 h prior to inoculation. This process would allow for assessments of a plant’s healing ability and not the disease prevention effect that this study demonstrated.
Conclusions and perspectives
Chlorine dioxide, in 50 mg L-1 doses, injected in the banana seedlings (Musa sp.) under greenhouse conditions significantly reduced the development of the Moko disease caused by R. solanacearum. In addition, the potential use of chitosan, methyl jasmonate and 3-aminobutanoic acid was observed for the management of the disease. Field tests must be performed to validate the results found in this study and to determine the applicability of the treatments that showed favorable management of R. solanacearum as possible alternative strategies for integrated management of the disease.
Acknowledgments
We would like to thank D. Ossa for previous tests of chlorine dioxide, J. Castaño for preparing the doses of the inducers and C. Gomez for assistance in the phytotoxicity tests. In addition, we would like to thank Cenibanano for providing the R. solanacearum strain. The present work was financed by the Universidad Nacional de Colombia, Medellin and Politecnico Colombiano Jaime Isaza Cadavid.
Literature cited
Agrios, G.N. 2005. Plant pathology. 5th ed. Elsevier; Academic Press, New York, NY.
Ait Barka, E., P. Eullaffroy, C. Clément, and G. Vernet. 2004. Chitosan improves development, and protects Vitis vinifera L. against Botrytis cinerea. Plant Cell Rep. 22, 608-614. Doi: 10.1007/s00299-003-0733-3
Arenas, A., D. López, E. Álvarez, G. Llano, and J. Loke. 2004. Efecto de prácticas ecológicas sobre la población de Ralstonia solanacearum Smith, causante de moko del plátano. Fitopatol. Colomb. 28, 76-80.
Augura, Asociación de Bananeros de Colombia. 2012. Coyun- tura bananera 2012. In: www.augura.com.co/index.php? option=com_docman&task=cat_view&gid=27&Itemid=95; consulted: May, 2015.
Buitrago G., E. 2001. Impacto socio-económico de la enfermedad del “Moko” en plantaciones de plátano y banano (Ralstonia solanacearum raza 2), en seis municipios del departamento del Quindío. pp. 31-35. In: Seminario-Taller Manejo Integrado de Sigatokas, “Moko” y Picudo Negro del Plátano en el Eje Cafetero. Sena; Corpoica, Armenia, Colombia.
Castañeda S., D.A. and J.A. Espinosa O. 2005. Comportamiento e impacto de la enfermedad de Moko en la zona de Urabá (Colombia), en las últimas tres décadas y media y propuesta de un índice de riesgo de la enfermedad. Rev. Fac. Nal. Agr. Medellin 58, 2587-2599.
Falcón-Rodríguez, A.B., D. Costales, J.C. Cabrera, and M.A. Martínez-Téllez. 2011. Chitosan physico-chemical properties modulate defense responses and resistance in tobacco plants against the oomycete Phytophthora nicotianae. Pest. Biochem. Physiol. 100, 221-228. Doi: 10.1016/j.pestbp.2011.04.005
French, E.B., L. Gutarra, P. Aley, and J. Elphinstone. 1995. Culture media for Ralstonia solanacearum isolation, identification and maintenance. Fitopatología 30, 126-130.
González, I., Y. Árias, and B. Peteira. 2009. Interacción planta-bac- terias fitopatógenas: caso de estudio Ralstonia solanacearum- plantas hospedantes. Rev. Protección Veg. 24, 69-80.
Herman, M.A.B., S. Restrepo, and C.D. Smart. 2007. Defense gene expression patterns of three SAR-induced tomato cultivars in the field. Physiol. Mol. Plant Pathol. 71, 192-200. Doi: 10.1016/j. pmpp.2008.02.002
Jakab, G., V. Cottier, V. Toquin, G. Rigoli, L. Zimmerli, J.-P. Métraux, and B. Mauch-Mani. 2001. β-aminobutyric acid-induced resistance in plants. Eur. J. Plant Pathol. 107, 29-37. Doi: 10.1023/A:1008730721037
Junli, H., W. Li, R. Nanqi, M. Fang, and J. Li. 1997. Disinfection effect of chlorine dioxide on bacteria in water. Water Res. 31, 607-613. Doi: 10.1016/S0043-1354(96)00275-8
Kelman, A. 1954. The relationship of pathogenicity in Pseudomonas solanacearum to colony appearance on a tetrazolium medium. Phytopathology 44, 693-695.
Madden, L.V., G. Hughes, and F. Van den Bosch. 2007. The study of plant disease epidemics. APS Press, St. Paul, MN.
Obregón B., M., P.A. Rodríguez G., J.G. Morales O., and M. Salazar Y. 2008. Hospedantes de Ralstonia solanacearum en planta- ciones de banano y plátano en Colombia. Rev. Fac. Nal. Agr. Medellin 61, 4518-4526.
Olivieri, F.P., M.C. Lobato, E. González Altamiranda, G.R. Daleo, M. Huarte, M.G. Guevara, and A.B. Andreu. 2009. BABA effects on the behaviour of potato cultivars infected by Phytophthora infestans and Fusarium solani. Eur. J. Plant Pathol. 123, 47-56. Doi: 10.1007/s10658-008-9340-z
Ozeretskovskaya, O.L., N.I. Vasyukova, N.G. Gerasimova, G.I. Chalenko, A.A. L’vova, A.N. Levov, A.V. Il’ina, V.P. Varlamov, and I.A. Tarchevskii. 2009. Potato resistance induced by chi- tosan derivatives. Dokl. Biol. Sci. 427, 355-357. Doi: 10.1134/ S0012496609040152
Prieto R., J., J.G. Morales O., and M. Salazar Y. 2012. Identification of new hosts for Ralstonia solanacearum (Smith) race 2 from Colombia. Rev. Protección Veg. 27, 151-161.
Ramírez J.R., A. Benjamid, and P.E. Buritica. 2014. Manejo de la Pudricion acuaso del pseudotallo (Dickeya sp.) en banano (Musa sp.) bajo condiciones de invernadero. Agron. Costarr. 38, 83-92.
Rodgers, S.L., J.N. Cash, M. Siddiq, and E.T. Ryser. 2004. A com- parison of different chemical sanitizers for inactivating Esch- erichia coli 0157H7 and Listeria monocytogenes in solution and in apple, lettuce, strawberries and cantaloupe. J. Food Prot. 67, 721-731.
Roldán, D., M. Salazar, M. Tejada, and Y. Peña. 2002. Caracterización de la cadena de plátano en Colombia. Ministerio de Agricultura y Desarrollo Rural (MADR), Bogota.
Sena. 2004. El plátano Musa spp. Su cosecha y postcosecha en la ca- dena agroindustrial. Asohofrucol; Fondo Nacional de Fomento Hortofrutícola; Corpoica; Gobernación del Meta; Corpometa, Villavicencio, Colombia.
Siegrist, J., M. Orober, and H. Buchenauer. 2000. β-Aminobutyric acid-mediated enhancement of resistance in tobacco to to- bacco mosaic virus depends on the accumulation of salicylic acid. Physiol. Mol. Plant Pathol. 56, 95-106. Doi: 10.1006/ pmpp.1999.0255
Silva, S. de O., S. de O Veras, L. Gasparotto, A.P. de Matos, Z.M. Cordeiro, and B. Boher. 2000. Evaluation of Musa spp. for resistance to moko disease (Ralstonia solanacearum, Raza 2). Infomusa 9, 19-20.
Singh, N., R.K. Singh, A.K. Bhunia, and R.L. Stroshine. 2002. Ef- ficacy of chlorine dioxide, ozone, and thyme essential oil or a sequential washing in killing Escherichia coli O157:H7 on lettuce and baby carrots. LWT- Food Sci. Technol. 35, 720-729. Doi: 10.1006/fstl.2002.0933
Smith, R. 1993. Polyclonal and monoclonal antibody-based enzyme- linked immunosorbent assays for Ralstonia solanacearum. p. 196. In: Proceedings of an International Workshop: Integrated Management of Bacteria Wilt. Indian Council of Agriculture Research, New Delhi.
Walters, D.R. 2009. Are plants in the field already induced? Implica- tions for practical disease control. Crop Prot. 28, 459-465. Doi: 10.1016/j.cropro.2009.01.009
Walters, D., D. Walsh, A. Newton, and G. Lyon. 2005. Induced resistance for plant disease control: maximising the efficacy of resistance elicitors. Phytopathology 95, 1368-1373. Doi: 10.1094/PHYTO-95-1368
Vallad, G.E. and R.M. Goodman. 2004. Systemic acquired resistance and induced systemic resistance in conventional agriculture. Crop Sci. 44, 1920-1934. Doi: 10.2135/cropsci2004.1920
Yin, Y., Y.-C. Li, Y. Bi, S.-J. Chen, Y.-C. Li, L. Yuan, Y. Wang, and D. Wang. 2010. Postharvest treatment with β-aminobutyric acid induces resistance against dry rot caused by Fusarium sulphureum in potato tuber. Agric. Sci. China 9, 1372-1380. Doi: 10.1016/S1671-2927(09)60228-5
Zhang, S. and J.M. Farber. 1996. The effects of various disinfectants against Listeria monocytogenes on fresh-cut vegetables. Food Microb. 13, 311-21. Doi: 10.1006/fmic.1996.0037
Zhu, Z. and S. Tian. 2012. Resistant responses of tomato fruit treated with exogenous methyl jasmonate to Botrytis cinerea infection. Sci. Hortic. 142, 38-43. Doi: 10.1016/j.scienta.2012.05.002

